Phys.org January 5, 2023
To learn more about how cancer cells size up surrounding tissue for stiffness and adapt their gecko-like movements in response a team of researchers at Virginia Polytechnic Institute developed a multiscale chemo mechanical whole-cell theory for mesenchymal migration. They developed a framework that coupled the subcellular focal adhesion dynamics at the cell-substrate interface with the cellular cytoskeletal mechanics and the chemical signaling pathways involving Rho GTPase proteins. In the presence of stiffness gradients and absence of chemical polarization, the cell moved toward an optimally stiff region from softer regions during durotaxis and from stiffer regions during anti-durotaxis. They showed that cell polarization through steep Rho GTPase gradients can reverse the migration direction dictated by the mechanical cues. Their theory demonstrated that opposing durotactic behaviors emerge via the interplay between intracellular signaling and cell-medium mechanical interactions in agreement with experiments, thereby elucidating complex mechanosensing at the single-cell level…read more. TECHNICAL ARTICLE
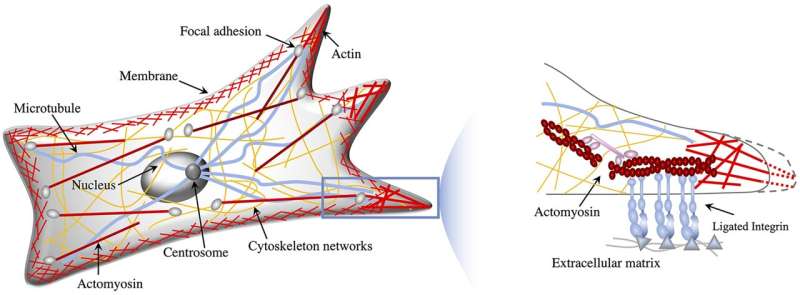
This schematic shows the intracellular organization that drives nucleus movement and mesenchymal migration… Credit: Wenya Shu