Phys.org August 29, 2019
Selective bond cleavage via vibrational excitation is the key to active control over molecular reactions. However, the practical implementation in condensed phases have been hampered to date by poor excitation efficiency due to fast vibrational relaxation. Researchers in Japan fabricated tiny gold antennae, each 300 nanometers wide, and illuminated them with infrared lasers. When infrared light of the right frequency was present, the electrons in the antennae oscillated back creating plasmonic resonance. The plasmonic resonance focused the laser’s energy on nearby molecules, which started vibrating. The vibration was further boosted by shaping the waveform of the infrared laser so that the frequency changed rapidly in time and broke the bond. This process may have applications throughout chemistry as a way to direct chemical reactions in desired directions, particularly in the energy, pharmaceutical sectors…read more. Open Access TECHNICAL ARTICLE
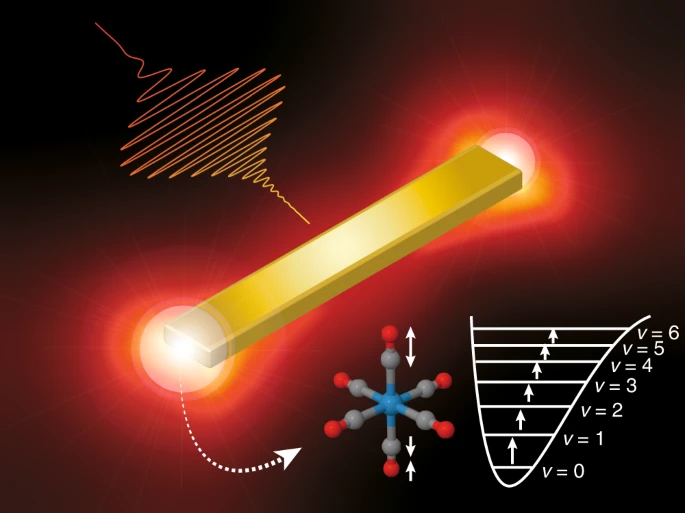
Schematic view of the antenna-enhanced vibrational ladder climbing. The chirped mid-IR pulse, enhanced with plasmon excitation of the gold nanoantenna, strongly excites the carbonyl stretch vibration of W(CO)6 molecules. Credit: Nature Communications (2019). DOI: 10.1038/s41467-019-11902-6